Revolutionizing Energy Storage: The Rise of Solid-State Batteries in the Electric Vehicle Era
Lithium-ion batteries commercially used are mainly composed of cathode, anode separator and electrolyte. Lithium-ion batteries are installed in smartphones, power tools, and EVs. They use liquid electrolyte solution.
Components of a battery-
Electrodes- The electrode that releases electrons during discharge is an anode; the electrode that absorbs the electrons is the cathode. They store lithium. The electrolyte carries positively charged lithium ions (i.e., an ion is an electrically charged particle produced to give a positive or negative ion by either removing or adding electrons from a neutral atom) from the anode to the cathode and vice versa through the separator. The movement of lithium ions creates free electrons in the anode, creating a charge that flows electric current through a device being powered. Electrodes are responsible for the basic performance of the battery.
Access the Complete report (including graphs, charts, and figures): https://univdatos.com/report/india-lithium-ion-battery-market/
Separator and Electrolyte- These two determine the safety of a battery. The separator acts as a physical barrier between cathode and anode, preventing the direct flow of electrons between them, blocking the flow of electrons inside the battery, leaving only ions to pass through them.
The electrolyte transfers ions (charge carrying particles) back and forth between the battery’s two electrodes, causing the battery to charge and discharge.
Solid-state battery- The solid-state battery uses a solid electrolyte solution instead of a liquid electrolyte solution. It also plays the role of a separator as well. It has a higher energy density. It does not have a risk of explosion or catching fire, removing the need to have safety components for those purposes, and saving more space for increasing battery capacity.
Example of Solid-State Battery
Experimentation with solid-state batteries traces back to the late 1950s, with the use of silver-ion conducting electrolytes. Fast forward to today, an example of a solid-state battery is lithium phosphate glass. The energy density is high in these batteries, with energy capacity greater than that of a Li-ion battery.
Advantages of Solid-State Battery
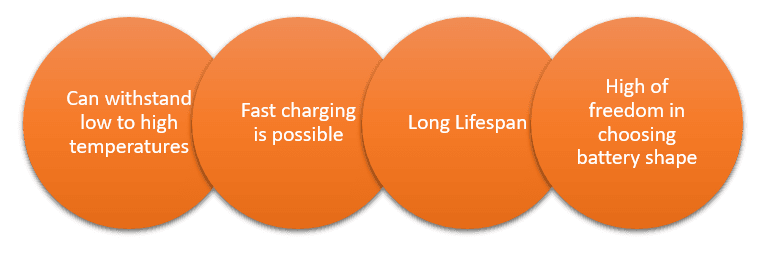
Liquid electrolytes in lithium-ion batteries are made of flammable organic solvents; there is a concern about their use in high-temperature environments. There is no such risk associated with the use of solid-state batteries, as they are not made of flammable materials. They can be used at high temperatures and make fast charging possible due to their heat resistance. Another benefit is the choice of battery size as there are no structural restrictions as in the case of liquid electrolytes to prevent leakages.
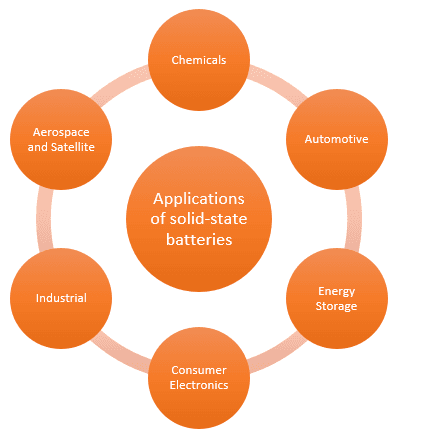
Role of Solid-State Batteries in Electric Vehicles
Research points towards the transition from ICEVs (Internal combustion electric vehicles) to electric vehicles in the auto industry. Several challenges need to be resolved before electric vehicles become mainstream. EVs need to have a similar level of mileage as the current ICEV, and it is necessary to increase the battery capacity of an EV to do so. Replacement of lithium-ion batteries with solid-state batteries is a major market trend in R&D for automotive and solid-state battery manufacturers.
The market size of the solid-state battery is expected to grow with a CAGR of 32.5%, reaching US$ 314 Million by 2028 from US$ 58 million in 2022.
Conclusion
Lithium-ion batteries are reaching their technological advancement. Solid-state batteries are a viable alternative to address current restraining factors. Huge investments by manufacturers (Toyota, BMW, Volkswagen, Hyundai and many more) in the R&D of solid-state batteries will result in innovative technological advancements in the sector.
Author: Abhishek Saini
