- Home
- About Us
- Industry
- Services
- Reading
- Contact Us
ERYTHROPOIETIN STIMULATING AGENTS MARKET SEEN SOARING 8.6% GROWTH TO REACH USD 15.2 BILLION BY 2030, PROJECTS UNIVDATOS
Author: Vikas Kumar
June 24, 2024
Key Highlights of the Report:
- Growing demand in the healthcare sector: The erythropoietin stimulating agent market is experiencing significant growth due to increasing demand in the healthcare sector, driven by growing concerns over widespread anemia cases.
- Rise in pharmaceutical market: There has been a growing demand for erythropoietin stimulating agents owing to increased incidences of cancer, and chronic kidney disorders leading to anemia.
- Major investments in the oncology sector: Pharmaceutical companies have adopted collaborative strategies to develop improved medications for treating oncology-related anemia.
According to a new report by UnivDatos, Erythropoietin stimulating agents (ESAs) are medications utilized to increase red blood cell production, often applied in managing anemia linked to conditions such as chronic kidney disease or cancer. They function akin to the natural hormone erythropoietin, prompting the bone marrow to generate more red blood cells. However, their use requires vigilant monitoring due to potential adverse effects like heightened blood clot risk or elevated blood pressure. Several factors are contributing to the growth of cancer cases including the increased cases of chronic kidney disorders, the surge in awareness programs regarding anemia, and rising investments in healthcare departments are driving the growth of the erythropoietin stimulating agent market. Apart from these factors, rising strategic collaborations are leading the market in this forecast period.
For More Detailed Analysis in PDF Format, Visit- https://univdatos.com/reports/erythropoietin-stimulating-agents-market?popup=report-enquiry
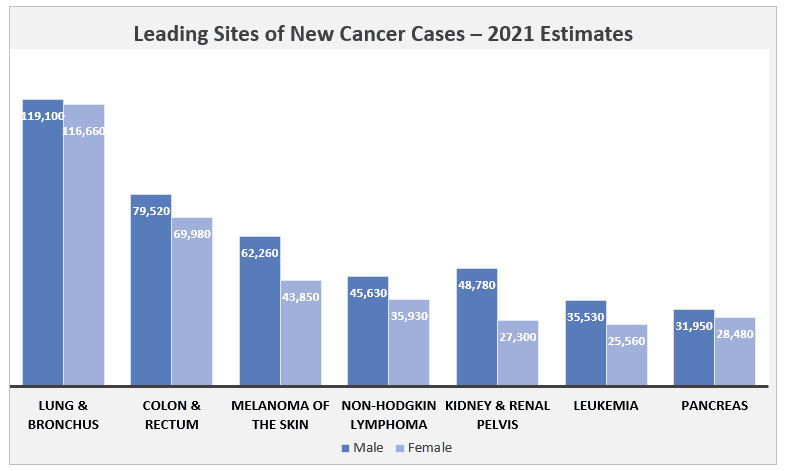
Fig: Leading Sites of New Cancer Cases – 2021 Estimates
The report suggests that the Rising Expenditure on the Oncology Sector is one of the major factors driving the growth of the erythropoietin stimulating agent market during the forthcoming years. Money is essential for gaining prominence in the market, and cancer therapeutics are gaining much of it. This expenditure is driving the market for the development of new technological advancements in anemia treatment. In recent years, there have been increasing clinical trials, a rising number of launches, and technological advancements focused on treating this blood deficiency. For instance, in July 2023, the University of Pittsburgh initiated a clinical trial for evaluating the safety and efficacy of erythropoietin (EPO) in combination with hydroxyurea in the treatment of chronic anemia in patients with sickle cell disease (SCD). It also provides hope for patients to get better care and support through erythropoietin stimulating agents, also it can serve as a doorway to opportunities for offering better treatment options in the future.
Darbepoetin-Alfa Gaining Maximum Traction in the Market
Darbepoetin alfa is among the erythropoietin-stimulating agents (ESAs) used to treat anemia. It was recognized for its longer half-life compared to other ESAs like epoetin alfa, allowing for less frequent dosing. Darbepoetin alfa is a synthetic form of erythropoietin, a hormone that stimulates red blood cell production in the bone marrow. It is used to treat anemia associated with chronic kidney disease, and chemotherapy in cancer patients, and to reduce the need for transfusions in certain surgeries. Darbepoetin alfa offers several advantages:
- Extended Half-Life: Its extended half-life allows for less frequent dosing compared to other erythropoietin-stimulating agents (ESAs), reducing the frequency of injections needed.
- Reduced Anemia Symptoms: By stimulating red blood cell production, it helps alleviate symptoms of anemia, such as fatigue and weakness, improving the overall quality of life for individuals with conditions like chronic kidney disease or undergoing chemotherapy.
- Minimized Need for Blood Transfusions: By boosting red blood cell production, darbepoetin alfa can decrease the need for blood transfusions, which is especially beneficial for patients with chronic kidney disease or certain types of cancer.
- Convenient Administration: Its less frequent dosing schedule can be more convenient for patients, potentially improving treatment adherence.
Conclusion
The global erythropoietin stimulating agent market is a rapidly growing field, with advancements in technology leading to improved outcomes for patients with the initial stage of the disease. The global erythropoietin stimulating agent market is expected to continue to grow in the coming years, as new technologies are developed. Overall, the global erythropoietin stimulating agent market represents a significant opportunity for pharmaceutical industries, which are making strategic alliances to design an effective drug for anemia treatment. With continued research and development, even more effective and personalized treatments will likely become available in the future, leading to improved outcomes for patients and doctors through erythropoietin-stimulating agents.
Key Offerings of the Report
Market Size, Trends, & Forecast by Revenue | 2023−2030
Market Dynamics – Leading Trends, Growth Drivers, Restraints, and Investment Opportunities
Market Segmentation – A detailed analysis by Product Type, Indication, and Region
Competitive Landscape – Top Key Vendors and Other Prominent Vendors
Get a Callback
Related News
Subscribe to our newsletters
By submitting this form, I understand that my data will be processed by Univdatos as indicated above and described in the Privacy Policy. *
