Biliary Tract Cancers Market: Hope on the Horizon
- Vikas Kumar
- March 16, 2024
- BLOG, HEALTHCARE
- Biliary Tract Cancers (BTCs) Market Forecast, Biliary Tract Cancers (BTCs) Market Growth, Biliary Tract Cancers (BTCs) Market Share, Biliary Tract Cancers (BTCs) Market Size, Biliary Tract Cancers (BTCs) Market Trends
- 0 Comments
Biliary tract cancers, including intrahepatic, perihilar, and distal cholangiocarcinoma as well as gallbladder cancer, are low-incidence malignancies in most high-income countries, but represent a major health problem in endemic areas; moreover, the incidence of intrahepatic cholangiocarcinoma is rising globally. Surgery is the cornerstone of cure; the optimal approach depends on the anatomical site of the primary tumor and the best outcomes are achieved through management by specialist multidisciplinary teams.
Unlock Insights: Receive a Sample Research Report on the Biliary Tract Cancers (BTCs) Market https://univdatos.com/get-a-free-sample-form-php/?product_id=42481
Biliary Tract Cancer Treatment
For all subtypes of CCA, surgical treatment is the only possible cure. However, the majority of CCA patients are diagnosed at late-stage disease, and nearly more than one-fourth of patients considered respectable are found to be unresectable during explorative laparotomy. Few more aggressive surgical approaches and improved radiologic techniques have resulted in improved R0 (tumor-free margins) resection rates, but recurrence rates remain high.
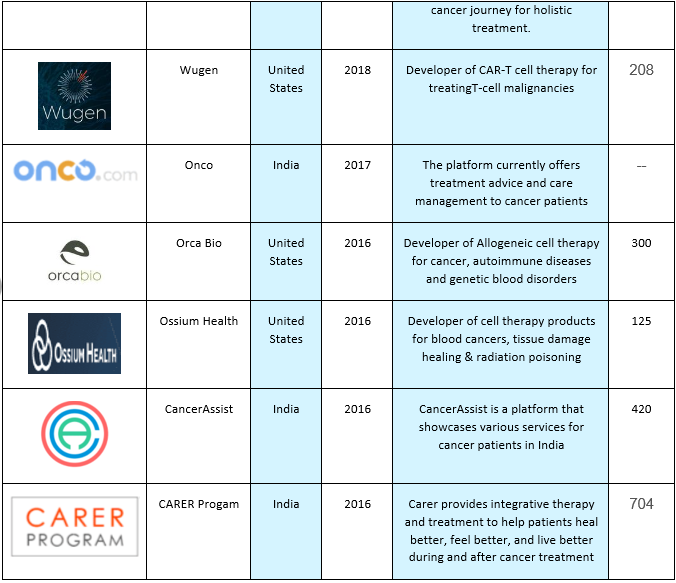
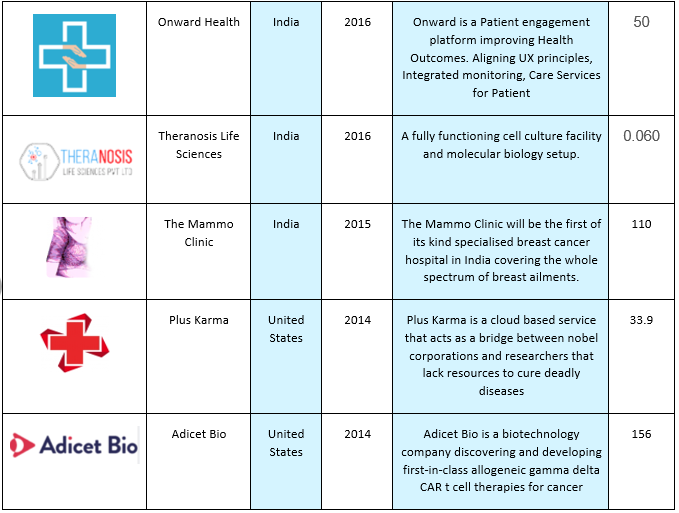
Here are some of the companies that are contributing towards Biliary tract cancers industry:
Current state of the biliary tract cancers industry
Recent Developments
In September 2022, Durvalumab (Imfinzi, AstraZeneca UK Limited) was granted approval by the Food and Drug Administration on September 2, 2022 for treating adult patients with locally advanced or metastatic biliary tract cancer (BTC) in combination with gemcitabine and cisplatin. In the TOPAZ-1 (NCT03875235) trial, which was a randomized, double-blind, placebo-controlled, multiregional study, the efficacy of the treatment was evaluated.
In June 2022, Heron Therapeutics Announced the Publication of ZYNRELEF as the Foundation of a Perioperative Non-Opioid Multimodal Analgesic Regimen Reduced Severe Pain and Opioid Use in Patients Having Total Knee Arthroplasty.
In March 2022, Heron Therapeutics obtained Pass-through Payment Status for ZYNRELEF from the Centers for Medicare & Medicaid Services, allowing for separate reimbursement in the hospital outpatient setting of care.
In May 2021, Amgen’s LUMAKRAS (sotorasib) was approved by the U.S. Food and Drug Administration (FDA) for the treatment of adult patients with KRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC).
Explore the Comprehensive Research Overview, Including a Table of Contents, on the Biliary Tract Cancers (BTCs) Market https://univdatos.com/report/biliary-tract-cancers-market/
Conclusion
While biliary tract cancers remain challenging to diagnose and treat, there is reason for hope. Advances in early detection, precision medicine, targeted therapies, immunotherapy, and minimally invasive surgery are improving outcomes for patients. The ongoing commitment of the medical community to research and innovation offers promise for a brighter future for those affected by these cancers. Patients, caregivers, and healthcare professionals should stay informed about the latest developments in the biliary tract cancers market to provide the best care possible. According to the UnivDatos Market Insights, the development of advanced technologies would massively transform the global scenario of biliary tract cancer and as per their “Biliary Tract Cancers (BTCs) Market” report, the global market was valued at USD~700 Mn in 2022, growing at a CAGR of 5.5% during the forecast period from 2023 – 2030.