- Home
- About Us
- Industry
- Services
- Reading
- Contact Us
Advanced Therapy Medicinal Products CDMO Market: Current Analysis and Forecast (2022-2028)
Emphasis on Product (Gene Therapy, Cell Therapy, Tissue Engineered, and Others); Phase (Phase I, Phase II, Phase III, and Phase IV); Indication (Oncology, Cardiology, Central Nervous System, Musculoskeletal, Infectious Disease, and Others); and Region/Country
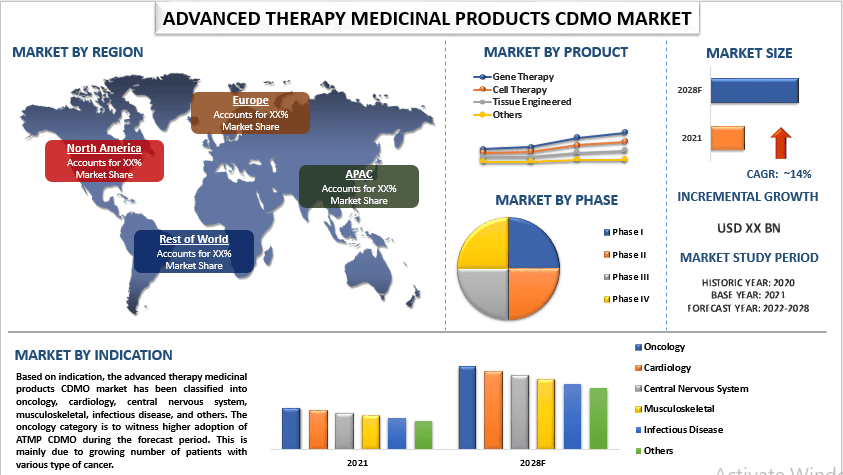
The global Advanced Therapy Medicinal Products CDMO Market is expected to grow at a significant rate of around 14% during the forecast period. Advanced Therapy Medicinal Products (ATMPs) are a new category of medicines that offer innovative treatments for various diseases and medical conditions. They include cell and gene therapies, tissue-engineered products, and other innovative treatments. The Contract Development and Manufacturing Organizations (CDMO) market for ATMPs provides services for the development and manufacturing of ATMPs, helping companies bring their products to market more quickly and cost-effectively.
The market for ATMPs is growing rapidly, driven by the increasing demand for new and effective treatments for various diseases and conditions, including cancer, genetic disorders, and chronic diseases such as diabetes and heart disease. The market is further driven by advancements in biotechnology and the increasing availability of cutting-edge technologies and the increasing demand for new and effective treatments for rare genetic diseases and conditions, the market is expected to continue growing in the coming years. For instance, as per the United States Center for Disease Control and Prevention article on spinal muscular atrophy (SMA) updated in December 2021, SMA is a genetic disorder that affects around 1 in every 10,000 people. Therefore, it is one of the most common rare diseases.
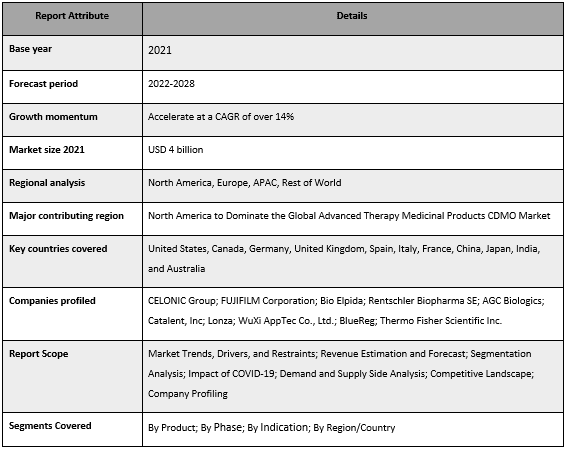
CELONIC Group; FUJIFILM Corporation; Bio Elpida; Rentschler Biopharma SE; AGC Biologics; Catalent, Inc; Lonza; WuXi AppTec Co., Ltd.; BlueReg; Thermo Fisher Scientific Inc. are some of the key players in the market. Several M&As along with partnerships have been undertaken by these players to facilitate customers with hi-tech and innovative products/technologies.
Insights Presented in the Report
“Amongst product, cell therapy category to witness fastest growth rate during the forecast period”
Based on product, the market is segmented into gene therapy, cell therapy, tissue engineered, and others. Amongst, cell therapy category to witness fastest growth rate during the forecast period. This segment includes the production and manufacturing of cellular products, such as stem cells, T-cells, and other cell-based treatments, which are used to treat a range of diseases and conditions, including cancer, genetic disorders, and degenerative diseases. Companies in this segment offer a range of services, including cell processing, cell banking, and cell therapy product development and commercialization. These factors are expected to contribute to the growth of this segment.
“Amongst indication, the cardiology category held a significant share in the market in 2021.”
On the basis of indication, the market is categorized into oncology, cardiology, central nervous system, musculoskeletal, infectious disease, and others. Among these, the cardiology category held a significant share in the market in 2021. This is mainly due to the several factors, including increasing prevalence of cardiovascular diseases such as heart attack, stroke, and heart failure, are leading cause of death and disability worldwide. The increasing prevalence of these diseases is driving the demand for new and innovative treatments, including ATMPs.
Advanced Therapy Medicinal Products CDMO Market Report Coverage
“APAC to hold a significant share in the market”
For a better understanding of the market adoption of the advanced therapy medicinal products CDMO industry, the market is analyzed based on its worldwide presence in the countries such as North America (U.S., Canada, Rest of North America), Europe (Germany, U.K., France, Spain, Italy, Rest of Europe), Asia-Pacific (China, Japan, India, Rest of Asia-Pacific), Rest of World. The ATMPs CDMO market in the APAC region is expected to continue growing in the coming years, driven by factors such as increasing demand for ATMPs, rising investment in the healthcare industry, and growing awareness about the benefits of these therapies.
The other factors driving the growth of this market in the region include the presence of several ATMP CDMOs in the APAC region such as large multinational corporations and local companies. The ATMP CDMO market in the region is characterized by high competition, with companies constantly looking to improve their production processes and capabilities to stay ahead of their competitors.
Reasons to buy this report:
- The study includes market sizing and forecasting analysis validated by authenticated key industry experts.
- The report presents a quick review of overall industry performance at one glance.
- The report covers an in-depth analysis of prominent industry peers with a primary focus on key business financials, product portfolio, expansion strategies, and recent developments.
- Detailed examination of drivers, restraints, key trends, and opportunities prevailing in the industry.
- The study comprehensively covers the market across different segments.
- Deep dive regional level analysis of the industry.
Customization Options:
The global advanced therapy medicinal products CDMO market can further be customized as per the requirement or any other market segment. Besides this, UMI understands that you may have your own business needs, hence feel free to connect with us to get a report that completely suits your requirements.
Table of Content
Research Methodology for the Advanced Therapy Medicinal Products CDMO Market Analysis (2022-2028)
Analyzing the historical market, estimating the current market, and forecasting the future market of the global advanced therapy medicinal products CDMO market were the three major steps undertaken to create and analyze the adoption of advanced therapy medicinal products CDMO in major regions globally. Exhaustive secondary research was conducted to collect the historical market numbers and estimate the current market size. Secondly, to validate these insights, numerous findings and assumptions were taken into consideration. Moreover, exhaustive primary interviews were also conducted, with industry experts across the value chain of the global advanced therapy medicinal products CDMO market. Post assumption and validation of market numbers through primary interviews, we employed a top-down/bottom-up approach to forecasting the complete market size. Thereafter, market breakdown and data triangulation methods were adopted to estimate and analyze the market size of segments and sub-segments of the industry pertains to. Detailed methodology is explained below:
Analysis of Historical Market Size
Step 1: In-Depth Study of Secondary Sources:
Detail secondary study was conducted to obtain the historical market size of the advanced therapy medicinal products CDMO market through company internal sources such as annual reports & financial statements, performance presentations, press releases, etc., and external sources including journals, news & articles, government publications, competitor publications, sector reports, third-party database, and other credible publications.
Step 2: Market Segmentation:
After obtaining the historical market size of the advanced therapy medicinal products CDMO market, we conducted a detailed secondary analysis to gather historical market insights and share for different segments & sub-segments for major regions. Major segments are included in the report as product, phase, and indication. Further country-level analyses were conducted to evaluate the overall adoption of testing models in that region.
Step 3: Factor Analysis:
After acquiring the historical market size of different segments and sub-segments, we conducted a detailed factor analysis to estimate the current market size of the advanced therapy medicinal products CDMO market. Further, we conducted factor analysis using dependent and independent variables such as various product, phase, and indications of advanced therapy medicinal products. A thorough analysis was conducted for demand and supply-side scenarios considering top partnerships, mergers and acquisitions, business expansion, and product launches in the advanced therapy medicinal products CDMO sector across the globe.
Current Market Size Estimate & Forecast
Current Market Sizing: Based on actionable insights from the above 3 steps, we arrived at the current market size, key players in the global advanced therapy medicinal products CDMO market, and market shares of the segments. All the required percentage shares split, and market breakdowns were determined using the above-mentioned secondary approach and were verified through primary interviews.
Estimation & Forecasting: For market estimation and forecast, weights were assigned to different factors including drivers & trends, restraints, and opportunities available for the stakeholders. After analyzing these factors, relevant forecasting techniques i.e., the top-down/bottom-up approach were applied to arrive at the market forecast for 2028 for different segments and sub-segments across the major markets globally. The research methodology adopted to estimate the market size encompasses:
- The industry’s market size, in terms of revenue (USD) and the adoption rate of the advanced therapy medicinal products CDMO market across the major markets domestically
- All percentage shares, splits, and breakdowns of market segments and sub-segments
- Key players in the global advanced therapy medicinal products CDMO market in terms of products offered. Also, the growth strategies adopted by these players to compete in the fast-growing market.
Market Size and Share Validation
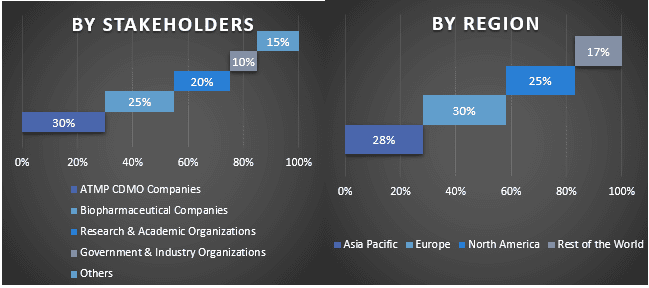
Primary Research: In-depth interviews were conducted with the Key Opinion Leaders (KOLs) including Top Level Executives (CXO/VPs, Sales Head, Marketing Head, Operational Head, Regional Head, Country Head, etc.) across major regions. Primary research findings were then summarized, and statistical analysis was performed to prove the stated hypothesis. Inputs from primary research were consolidated with secondary findings, hence turning information into actionable insights.
Split of Primary Participants in Different Regions
Market Engineering
The data triangulation technique was employed to complete the overall market estimation and to arrive at precise statistical numbers for each segment and sub-segment of the global advanced therapy medicinal products CDMO market. Data was split into several segments & sub-segments post studying various parameters and trends in the areas of product, phase, and indication in the global advanced therapy medicinal products CDMO market.
The main objective of the Global Advanced Therapy Medicinal Products CDMO Market Study
The current & future market trends of the global advanced therapy medicinal products CDMO market were pinpointed in the study. Investors can gain strategic insights to base their discretion for investments on the qualitative and quantitative analysis performed in the study. Current and future market trends determined the overall attractiveness of the market at a regional level, providing a platform for the industrial participant to exploit the untapped market to benefit from a first-mover advantage. Other quantitative goals of the studies include:
- Analyze the current and forecast market size of the advanced therapy medicinal products CDMO market in terms of value (USD). Also, analyze the current and forecast market size of different segments and sub-segments
- Segments in the study include areas of product, phase, and indication.
- Define and analysis of the regulatory framework for the advanced therapy medicinal products
- Analyze the value chain involved with the presence of various intermediaries, along with analyzing customer and competitor behaviors of the industry.
- Analyze the current and forecast market size of the advanced therapy medicinal products CDMO market for the major region.
- Major countries of regions studied in the report include Asia Pacific, Europe, North America, and the Rest of the World.
- Company profiles of the Advanced Therapy Medicinal Products CDMO Market and the growth strategies adopted by the market players to sustain in the fast-growing market.
- Deep dive regional level analysis of the industry
Related Reports
Customers who bought this item also bought