- Home
- About Us
- Industry
- Services
- Reading
- Contact Us
Point of Care Testing Market: Current Analysis and Forecast 2019-2025
Emphasis on Product (Blood Glucose Monitoring Test, Pregnancy & Fertility Testing, Infectious Disease Testing, Cardiac Markers, Anticoagulant Management, Others), Prescription (Near the Patient Testing (NPT), Over the Counter (OCT) /Patient Self-Testing (PST)), End-User (Hospitals, Clinics & Laboratory, Home Setting, Other Health Facilities) and Regions/countries
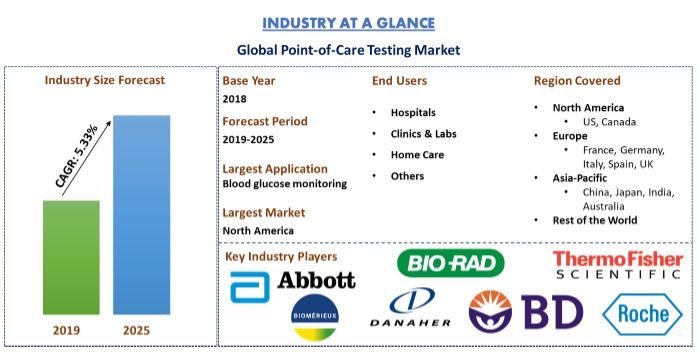
Global Point-of-Care Testing Market (POCT) was valued at US$ 20.42 billion in 2018 and is anticipated to reach US$ 29.27 billion by 2025 displaying a reasonable CAGR of 5.33% over the forecast period (2019-2025). With emerging technological advancements in healthcare, including lab-on-a-chip, smartphone apps, biosensors, and wearable devices that offer a closer connection to the patient point-of-care (POC) technologies are swiftly becoming part of the transformation of the healthcare landscape. The guiding concept in support of Point-of-Care Testing (POCT) is to bring testing closer to the patient. POCT offers more flexibility to meet a diverse range of medical needs because small, portable POCT devices make testing possible in remote locations, such as underserved populations, rural areas, and locations with limited infrastructure or personnel such as disaster, accident, or military sites. The market of POCT is anticipated to grow enormously owing to factors such as rising old age population and age-related diseases, technological advancements. Moreover, development of condition-specific markers and tests and growing significance of Companion Diagnostic will further boost this market. Growing significance of companion diagnostics and investing in unifying technology will further boom the industry and will mark as an upcoming trend. However, stringent government regulations, unfavourable reimbursement policies for POCT and risk of errors would act as the major challenges, but at a low level.
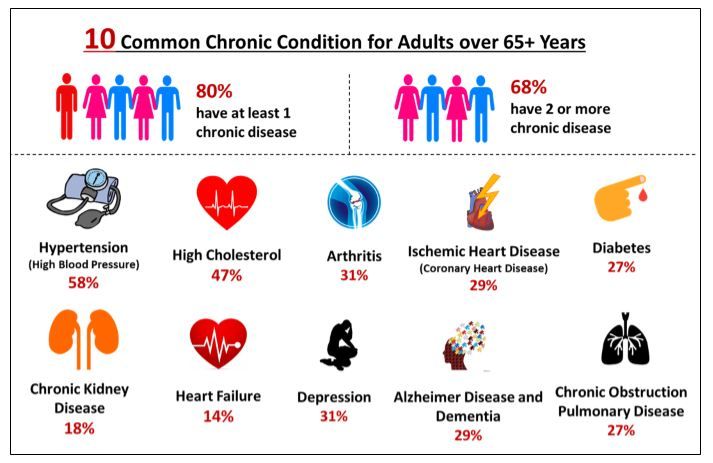
Increasing Prevalence of Chronic Diseases paired with Rising Geriatric (Elderly) Population is the major factor driving the adoption of POCT service. Costly chronic care needs are growing and exerting considerable demand for health systems. Chronic diseases are among the most prevalent and costly health conditions in the United States. Nearly half (approximately 45%, or 133 Million) of all Americans suffer from at least one chronic disease, and the number is growing. Chronic diseases including, cancer, diabetes, hypertension, stroke, heart disease, respiratory diseases, arthritis, obesity, and oral diseases can lead to hospitalization, long-term disability, reduced quality of life, and death. In fact, persistent conditions are the nation’s leading cause of death and disability.
“Amongst POCT product type, blood glucose testing products dominated the market accounting for 75.1% share in 2018”
Based on POCT product, the report segments the market into, Blood Glucose Monitoring Test, Pregnancy & Fertility Testing, Infectious Disease Testing, Cardiac Markers, Anticoagulant Management, Others. Blood Glucose Monitoring Test products dominated the market and is expected to maintain its dominance throughout the forecast period 2019-2025. However, Infectious Disease Testing products are expected to witness the highest CAGR during the analyzed period.
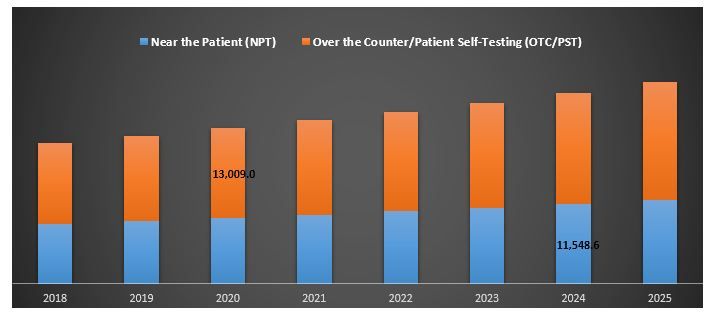
“Over the Counter/Patient Self-Testing (OTC/PST) dominated the market in 2018”
Based on prescription type the global POCT market is segmented into Near the Patient (NPT) and Over the Counter (OTC) /Patient Self-Testing (PST). Over the Counter/Patient Self-Testing (OTC/PST) occupied the largest share and is expected to maintain its dominance throughout the forecast period 2019-2025. Lack of time among the individuals paired with increased disposable income and technological advancement would be the major driving factor for the growth of the segments
Point-of-Care Testing Market by Prescription, Global, 2018-2025 (US$ Million)
“Hospitals were the major end-user of POCT technology. The segment held 47.7% share in 2018”
On the basis of end-users, the report segments the global POCT market into Hospitals, Clinics & laboratories, home settings and others are the major end-users. In 2018, Hospitals were the largest revenue-generating segment for technology. However, increasing elderly population and associated diseases would drive the increased adoption of POCT for home setting, hence the segment is expected to witness highest CAGR growth of 6.09% during the forecast period 2019-2025.
“North America represents the largest markets for POCT technology globally in 2018, followed by Asia-Pacific and Europe”
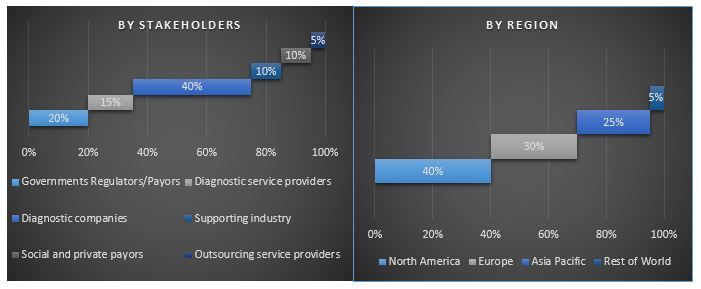
For a deep-dive analysis of the overall adoption of POCT technology, detailed analysis was conducted for major regions/countries including North America (US, Canada), Europe (Germany, France, Spain, Italy, UK), Asia-Pacific (China, India, Japan, Australia) and Rest of the world. In 2018, North America dominated the market with ~half the market. However, increasing elderly populations majorly in countries such as Japan and China, Asia-Pacific would emerge as one of the larger markets for the technology in years to come.
Competitive Landscape-Top 10 Market Players
Roche Diagnostics, Becton, Dickinson and Company, Abbott Laboratories, Inc., Biomérieux SA, Bio-Rad Laboratories, Inc., Danaher Corporation, Johnson & Johnson, Siemens Healthineers, Sysmex Corporation and Thermo Fisher Scientific are some of the prominent players operating in the global point-of-care testing market at global scale. However, owing to increasing old age population and rising prevalence age-related disorders, the industry has witnessed emergence of numerous start-ups operating at regional/country level.
Reasons to buy:
- Current and future market size from 2018 to 2025 in terms of value (US$)
- Combined analysis of deep-dive secondary research and input from primary research through Key Opinion Leaders of the industry
- Country-level details of the overall adoption of point of care market along with value chain
- A quick review of overall industry performance at a glance
- In-depth analysis of key industry players
- A detailed analysis of regulatory framework, drivers, restraints, key trends and opportunities, value chain prevailing in the industry
- Examination of industry attractiveness with the help of Porter’s Five Forces analysis
- The study comprehensively covers the market across different segments and sub-segments of the technology
- Region/country Covered: North America (US, Canada and Rest of North America), Europe (UK, Germany, France, Italy, Spain, Rest of Europe), Asia-Pacific (China, India, South Korea, Japan, Australia and Rest of Asia-Pacific), and Rest of the World
Customization Options:
The Global Point-of-Care testing Market can be customized to the country level or any other application. Besides this, UMI understands that you may have your own business need, hence we also provide fully customized solutions to clients. The Global POCT Market can be customized to the country level or any other market segment.
Table of Content
Analyzing historical market, estimation of the current market and forecasting the future market for Global POCT market were the three major steps undertaken to create and analyze the overall adoption rate of POCT services in major markets. Exhaustive secondary research was conducted to collect the historical market of the technology and overall estimation of the current market. Secondly, to validate these insights, numerous findings and assumptions were taken into consideration. Moreover, exhaustive primary interviews were conducted with industry experts across value chain of the POCT technology. Post all the assumptions and validation of market numbers through primary interviews, top-down approach was employed to forecast the complete market size of POCT technology at Global scale. Thereafter, market breakdown and data triangulation methods were adopted to estimate and analyze the overall market size of segments and sub-segments of the technology. Detailed methodology is explained below:
Analysis of Historical Market Size
Step 1: In-Depth Study of Secondary Sources:
Detail secondary study was conducted to obtain the historical market size of the Global POCT Market through company internal sources such as annual report & financial statements, performance presentations, press releases, inventory records etc. and external sources including trade journals, news & articles, government publications, economic data, competitor publications, sector reports, regulatory bodies publications, safety standard organizations, third-party database and other credible publications. For economic data collection, sources such as World Bank, CDC, European Commission (EC), United Nations and WHO, OECD among others were used.
Step 2: Market Segmentation:
Post obtaining the historical market size of the overall market, detailed secondary analysis was conducted to gather historical market insights and share for different segments and sub-segments of the Global POCT service. Major segments included in the report are product, prescription and end-user.
Step 3: Factor Analysis:
After acquiring the historical market size of different segments detailed factor analysis was conducted to estimate the current market size of the Global POCT technology. Factor analysis was conducted using dependent and independent variables such as Accelerating aging population and rising prevalence of age-related disorders, Technology Advancements, etc. Historical trends of the Global POCT and their year-on-year impact on the market size and share in the recent past were analyzed. The demand and supply-side scenario were also thoroughly studied.
Current Market Size Estimate & Forecast
Current Market Sizing: Based on actionable insights from the above 3 steps, we arrived at current market size, key players in major applications and markets, market shares of these players and industry’s supply chain. All the required percentage shares split, and market breakdowns were determined using the above-mentioned secondary approach and were verified through primary interviews.
Estimation & Forecasting: For market estimation and forecast, weightage was assigned to different factors including market dynamics such as drivers, restraints, and trends. After analyzing these factors, relevant forecasting techniques i.e. Bottom-up/Top-down was applied to arrive at the market forecast pertaining to 2025 for different segments and sub-segments in major markets. The research methodology adopted to estimate the market size encompasses:
- The industry’s market size, in terms of value and rate of adoption of POCT worldwide
- All percentage shares, splits and breakdowns of market segments and sub-segments
- Key players in major applications and markets as well as the market share of each player. Also, the growth strategies adopted by these players to compete in the ever-growing Global POCT market
Market Size and Share Validation
Primary Research: In-depth interviews were conducted with the Key Opinion Leaders (KOLs) including Top Level Executives (CXO/VPs, Sales Head, Marketing Head, Operational Head, and Regional Head, etc.). Primary research findings were summarized, and statistical analysis was performed to prove the stated hypothesis. Input from primary research was consolidated with secondary findings, hence turning information into actionable insights.
Split of Primary Participants
Market Engineering
Data triangulation technique was employed to complete the overall market engineering process and to arrive at precise statistical numbers of each segment and sub-segment pertaining to the Global POCT market. Data was split into several segments post studying various parameters and trends in the areas of disease testing such as Blood Glucose Monitoring Test, Pregnancy & Fertility Testing, Infectious Disease Testing, Cardiac Markers, Anticoagulant Management and others. A detailed analysis was also conducted for major regions including North America, Europe, Asia-Pacific among others
Main objective of the Global Point-of-Care Testing (POCT) Market Study
The current & future market trends of the global POCT market are pinpointed in the study. The investors can gain strategic insights to base their discretion for investments from the qualitative and quantitative analysis performed in the study. The current and future market trends would determine the overall attractiveness of the market, providing a platform for the diagnostics healthcare participant to exploit the untapped market to benefit as the first-mover advantage. Other quantitative goals of the studies include:
- Analyze the current and forecast market size of the global Point-of-Care Testing market in terms of value (US$)
- Analyze the current and forecast market size of different segments and sub-segments of the market
- To analyze the revenue and business models of the major market players in the industry along with their growth strategy adopted to sustain in the competitive market
- To understand the initiatives undertaken by diagnostics companies’ vs service providers to increase the overall analysis of Point-of-Care Testing in major regions
- Define and describe the segments and sub-segments considered in the evaluation of the global Point-of-Care Testing market and anticipate potential risk associated with the market
- Define and analysis of the government regulations for diagnostics and point-of-care testing industry
- Analyze the current and forecast market size of the market for major regions/countries including North America (US, Canada, Rest of North America), Europe (Germany, France, Spain, Italy, UK, Rest of Europe), Asia-Pacific (China, Japan, India, Australia and Rest of Asia-Pacific) and Rest of World
- Define and analyze the competitive landscape of the global Point-of-Care Testing market and the growth strategies adopted by the market players to sustain in the fast-growing market
Related Reports
Customers who bought this item also bought