- Home
- About Us
- Industry
- Services
- Reading
- Contact Us
Clinical Trials Market: Current Analysis and Forecast (2021-2027)
Emphasis on Phase (Phase I, Phase II, Phase III, and Phase IV); Study Design (Interventional Study, Observational Study, and Expanded Access Study); Therapeutic Area (Oncology, Clinical Pharmacology, Cardiology, Infectious Disease, Neurology, Gastroenterology & Hepatology, Ophthalmology, and Others)); Region/Country
The Global Clinical Trials Market is anticipated to grow with an elevated CAGR of around 8% over the forecast period (2021-2027). Clinical Trials are the important phases in the new drug development phase in which drug trials are conducted on human subjects to test the study of the possible side effects and to determine the dose of the medicine.
The growth of Clinical Trials market across the globe is witnessing growth on account of the rising prevalence of various chronic and rare diseases which leads to the development of new and more effective drugs for the disease. As per the national policy on rare diseases, the 7000 – 8000 rare diseases, but less than 5% have therapies available to treat them. About 95% of rare diseases have no approved treatment and less than 1 in 10 patients receive disease-specific treatment. Where drugs are available, they are prohibitively expensive, placing immense strain on resources. Additionally, the advancement in bioinformatics and the development of AI-based software that provide a better platform for artificial drug testing along with the development of large databases for drug testing during the different phases of the clinical trials and lead compound significantly contribute to the market growth of the clinical trial market.
IQVIA, PAREXEL International Corporation, ICON Plc, PRA Health Sciences, Syneos Health, Eli Lilly and Company, Novo Nordisk A/S, Pfizer, and Clinipace., are some of the prominent players operating in the Clinical Trial market. Several M&As along with partnerships have been undertaken by these players to facilitate customers with new varieties of Clinical Trial devices.
Insights Presented in the Report
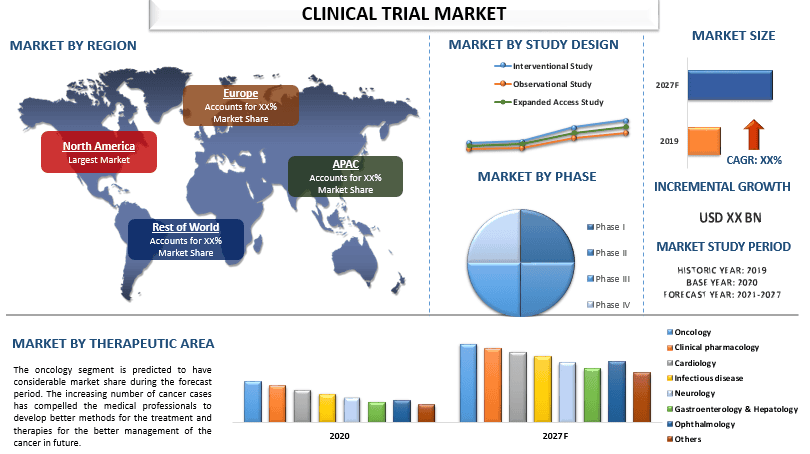
“Amongst Phase, Phase iii segment holds the major share”
Based on phase, the market is segmented into phase I, phase ii, phase iii, and phase iv. The phase iii segment held a significant market share in 2020. The significant share of the segment is due to the involvement of numerous subjects during the trials. Furthermore, this step is generally the last step before the licensing. The phase ii segment is also expected to have a lucrative share in the clinical trial market. As per FDA, a few hundred volunteers participate in these studies and around 33% of the drugs are passed to the next phase during this phase, the efficacy and are tested.
“Amongst Study design, interventional study segment holds the major share”
Based on the study design, the clinical trials market is divided into interventional study, observational study, and expanded access study. The interventional study segment grabbed a significant market share in 2020 and is anticipated to grow at a rapid rate in the upcoming years owing to the accurate observations taken in these studies and after the successful interventional studies, the observational and expanded access studies are conducted. Furthermore, the increasing population of people having prior medical conditions and the surging geriatric population coupled with the rising prevalence of multiple disorders requires the close monitoring of the diseases of this population which in turn increases the market size of the segment in the clinical trial market. For instance, as per the WHO, between 2015 and 2050, the proportion of the world’s population over 60 years will nearly double from 12% to 22%. Also, 1 in 6 people in the world will be aged 60 years or over by 2030.
“Amongst Therapeutic area, oncology segment holds the major share”
Based on the therapeutic area, the clinical trials market is divided into oncology, clinical pharmacology, cardiology, infectious disease, neurology, gastroenterology & hepatology, ophthalmology, and others. The oncology segment grabbed a significant market share in 2020 and is anticipated to grow at a rapid rate in the upcoming years owing to the increasing number of cancer cases which force medical professionals to develop new and effective drugs for the treatment of cancer. As per WHO, Cancer is a leading cause of death worldwide, accounting for nearly 10 million deaths in 2020. Among them, lung and colon cancers were the leading causes of death.
“North America represents one of the largest markets of Clinical Trial market”
For a better understanding of the market dynamics of the Clinical Trial market, a detailed analysis was conducted for different regions across the globe including North America (U.S, Canada, and the Rest of North America), Europe (Germany, France, Spain, United Kingdom, Italy, and Rest of Europe), Asia-Pacific (China, India, Australia, Japan, and Rest of APAC), Rest of World has been conducted. In 2020, North America grabbed a significant market share of the global clinical trials market 2020, and it is expected to grow with considerable CAGR during the forecast period (2021-2027F). The major factors such as favorable government initiatives, sophisticated healthcare, and research infrastructure, and the rising incidence of rare diseases across the region are propelling the growth of this market along with the rising research and development investment in the region. For instance, the Research and development (R&D) expenditures of Canada in 2019 reached $40.3 billion, a 3.9% increase from 2018. However, in the next year, the investment decreased due to the COVID-19 pandemic.
Reasons to buy this report:
- The study includes market sizing and forecasting analysis validated by authenticated key industry experts
- The report presents a quick review of overall industry performance at one glance
- The report covers an in-depth analysis of prominent industry peers with a primary focus on key business financials, product portfolio, expansion strategies, and recent developments
- Detailed examination of drivers, restraints, key trends, and opportunities prevailing in the industry
- The study comprehensively covers the market across different segments
- Deep dive regional level analysis of the industry
Customization Options:
The global Clinical Trial market can further be customized as per the requirement or any other market segment. Besides this, UMI understands that you may have your own business needs, hence feel free to connect with us to get a report that completely suits your requirements.
Table of Content
Research Methodology for the Global Clinical Trial Market Analysis (2021-2027)
Analyzing the historical market, estimating the current market, and forecasting the future market of the global Clinical Trial market were the three major steps undertaken to create and analyze the adoption of clinical trials in major regions globally. Exhaustive secondary research was conducted to collect the historical market numbers and estimate the current market size. Secondly, to validate these insights, numerous findings and assumptions were taken into consideration. Moreover, exhaustive primary interviews were also conducted, with industry experts across the value chain of the global clinical trial market. Post assumption and validation of market numbers through primary interviews, we employed a top-down/bottom-up approach to forecasting the complete market size. Thereafter, market breakdown and data triangulation methods were adopted to estimate and analyze the market size of segments and sub-segments of the industry pertains to. Detailed methodology is explained below:
Analysis of Historical Market Size
Step 1: In-Depth Study of Secondary Sources:
Detail secondary study was conducted to obtain the historical market size of the clinical trial through company internal sources such as annual reports & financial statements, performance presentations, press releases, etc., and external sources including journals, news & articles, government publications, competitor publications, sector reports, third-party database, and other credible publications.
Step 2: Market Segmentation:
After obtaining the historical market size of the clinical trial market, we conducted a detailed secondary analysis to gather historical market insights and share for different segments & sub-segments for major regions. Major segments are included in the report as product type and application. Further country-level analyses were conducted to evaluate the overall adoption of clinical trials across the globe.
Step 3: Factor Analysis:
After acquiring the historical market size of different segments and sub-segments, we conducted a detailed factor analysis to estimate the current market size of the clinical trial. Further, we conducted factor analysis using dependent and independent variables such as the growing number of people with chronic diseases and the increasing elderly population all over the globe. A thorough analysis was conducted for demand and supply-side scenarios considering top partnerships, mergers and acquisitions, business expansion, and product launches in the clinical trial sector across the globe.
Current Market Size Estimate & Forecast
Current Market Sizing: Based on actionable insights from the above 3 steps, we arrived at the current market size, key players in the clinical trial market, and market shares of the segments. All the required percentage shares split, and market breakdowns were determined using the above-mentioned secondary approach and were verified through primary interviews.
Estimation & Forecasting: For market estimation and forecast, weights were assigned to different factors including drivers & trends, restraints, and opportunities available for the stakeholders. After analyzing these factors, relevant forecasting techniques i.e., the top-down/bottom-up approach were applied to arrive at the market forecast for 2027 for different segments and subsegments across the major markets globally. The research methodology adopted to estimate the market size encompasses:
- The industry’s market size, in terms of value (US$) and the adoption rate of the clinical trials across the major markets domestically
- All percentage shares, splits, and breakdowns of market segments and sub-segments
- Key players in the clinical trial market in terms of products offered. also, the growth strategies adopted by these players to compete in the fast-growing market
Market Size and Share Validation
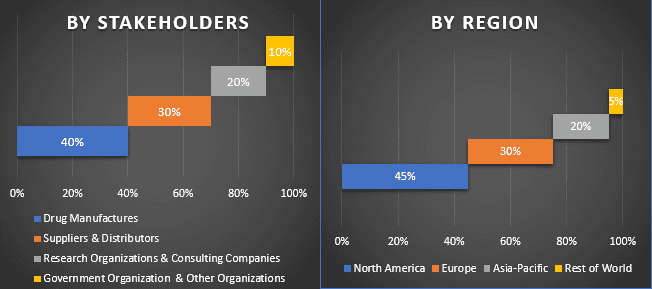
Primary Research: In-depth interviews were conducted with the Key Opinion Leaders (KOLs) including Top Level Executives (CXO/VPs, Sales Head, Marketing Head, Operational Head, Regional Head, Country Head, etc.) across major regions. Primary research findings were then summarized, and statistical analysis was performed to prove the stated hypothesis. Inputs from primary research were consolidated with secondary findings, hence turning information into actionable insights.
Split of Primary Participants in Different Regions
Market Engineering
The data triangulation technique was employed to complete the overall market estimation and to arrive at precise statistical numbers for each segment and sub-segment of the clinical trial market. Data was split into several segments & sub-segments post studying various parameters and trends in the areas of phase, study design, and therapeutic area of the clinical trial market.
The main objective of the clinical trial market study
The current & future market trends of clinical trials were pinpointed in the study. Investors can gain strategic insights to base their discretion for investments on the qualitative and quantitative analysis performed in the study. Current and future market trends determined the overall attractiveness of the market at a regional level, providing a platform for the industrial participant to exploit the untapped market to benefit from a first-mover advantage. Other quantitative goals of the studies include:
- Analyze the current and forecast market size of clinical trials in terms of value (US$). Also, analyze the current and forecast market size of different segments and sub-segments
- Segments in the study include areas of phase, study design, and therapeutic area
- Define and analysis of the regulatory framework for the clinical trial industry
- Analyze the value chain involved with the presence of various intermediaries, along with analyzing customer and competitor behaviors of the industry
- Analyze the current and forecast market size of the clinical trial market for the major region
- Major regions studied in the report include North America, Europe, Asia-Pacific, and Rest of the world
- Company profiles of the clinical trial market and the growth strategies adopted by the market players to sustain in the fast-growing market
- Deep dive regional level analysis of the industry
Related Reports
Customers who bought this item also bought